STEMart, a US-based provider of comprehensive services for all phases of medical device development, announced the launch of its new Force Degradation Services to help pharmaceutical and medical device companies evaluate the stability of their drug candidates and finished products under a variety of stress conditions, ensuring their safety and efficacy throughout their shelf life.
Forced degradation studies are used to demonstrate the molecular chemical behavior of processed products that may undergo degradation. These studies are typically performed prior to final formulation as they provide a rapid screening of material stability under external stress. It is important to note that in forced degradation studies, the degradation products generated in the stressed sample are referred to as “potential” degradation products, which may or may not be formed under the relevant storage conditions.
STEMart now offers various Forced Degradation Services, including hydrolytic degradation, oxidative degradation, photolytic degradation and thermolytic degradation. STEMart can provide comprehensive services to assist manufacturers in meeting regulatory goals and minimize compliance risk through extensive expertise.
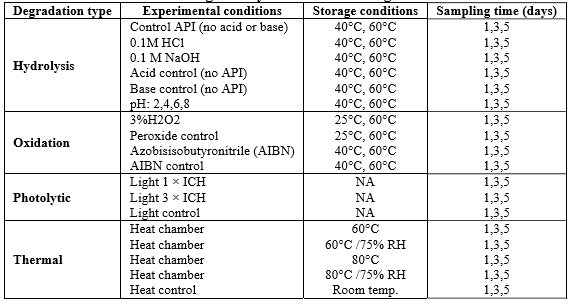
Degradation by hydrolysis is hydrolytic degradation. Hydrolysis reactions are usually catalyzed by acids or bases. STEM recommends that hydrolytic degradation be performed at temperatures around 70 degrees Celsius, with a reflux condenser installed to avoid evaporation losses and a small number of glass beads or ceramic tiles used to avoid collisions, and that stress reflux always be performed in a fume hood.
Oxidative degradation is one of the most common mechanisms of drug degradation and is usually performed at room temperature under dark conditions. It is not recommended to perform it at higher temperatures because at higher temperatures the rate of reaction in solution may actually decrease due to the reduction of oxygen content in the solvent. Typical Stress Conditions: 24 hours exposure to 3% hydrogen peroxide or 5% to 20% degradation, whichever comes first.
Photolytic degradation is degradation that results from exposure to UV or visible light. Typical exposure conditions for this type of forced degradation study are three exposures to 1.2 million lux-hours of visible light and 200 watt-hours per square meter of UV light.
Pyrolytic degradation studies include various reactions such as pyrolysis, hydrolysis, decarboxylation, isomerization, rearrangement, and polymerization. These reactions determine the melting point of the analyte. The general procedure is to stress and monitor the samples for one week, during which time the samples are tested to achieve a degradation rate of 5% to 20%, and the molecule is considered stable if no degradation occurs after more severe stress is applied.
If you have additional questions about Force Degradation Services or would like to find out more about Medical Device Testing services, please visit https://www.ste-mart.com/forced-degradation.htm.
About STEMart
STEMart is an industry-leading eCommerce platform incorporated with an extensive global footprint and a broad portfolio of more than 10,000 products. It aims to provide better lab materials, medical instruments and consumables, excellent technologies, and high-quality services to global customers in the fields of science, technology, and engineering, from the discovery stage upward to the manufacturing process. STEMart is dedicated to enhancing research and biotech production with simpler and safer protocols to access better health worldwide.




